Shanghai Vitalgen BioPharma Co., Ltd. (hereinafter referred to as "Vitalgen" or the "Company") recently announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has agreed to grant PRIME designation for VGR-R01 for the treatment of Bietti's crystalline dystrophy (BCD), supporting its development and accelerated assessment. The public information is as follows:

Source: EMA Website
The inclusion of VGR-R01 in the PRIME scheme is primarily based on evidence from Phase I/II clinical studies, in which two-thirds (2/3) of patients treated with VGR-R01 demonstrated significant improvement in best-corrected visual acuity (BCVA) (≥ -0.3 logMAR), with no patients experiencing significant worsening of visual acuity. The CHMP acknowledged that VGR-R01 has demonstrated clinical results with the potential for significant advantage and the ability to address an unmet medical need, thereby granting and publicly announcing the designation. Subsequently, VGR-R01 will benefit from proactive scientific advice and regulatory guidance from the EMA, as well as support in optimizing the development plan to ensure that study data are suitable for evaluation in a Marketing Authorization Application (MAA) in Europe, thereby improving the efficiency of drug development and review. Additionally, the product is eligible for accelerated assessment upon submission of a marketing authorization application in Europe, expediting the path to market.
Dr. Zhao Xiaoping, CEO of the company, stated, "The granting of PRIME designation for VGR-R01 by the European Medicines Agency represents authoritative recognition from an international regulatory body of the product's clinical value and marks another significant milestone in the Company's journey toward globalization. We will actively leverage the policy support provided by this acceleration program and maintain close communication with the Committee for Advanced Therapies (CAT) to efficiently advance the European marketing process for VGR-R01, aiming to fill the global therapeutic gap in BCD and bring benefits to patients worldwide with BCD as soon as possible. "
About the PRIME Scheme
PRIME (PRIority MEdicines) is an acceleration program launched by the EMA in 2016. It aims to support the development of medicines that address unmet medical needs by enhancing interaction with applicants, providing strengthened scientific guidance, and optimizing development plans so that generated data meet the requirements for a Marketing Authorization Application (MAA), ultimately enabling patients to benefit earlier from promising therapies. PRIME designation takes effect immediately upon granting. The EMA will proactively appoint rapporteurs from the CHMP or CAT to provide scientific and regulatory advice on the overall development plan and key milestones for the priority medicine. Furthermore, there are opportunities to involve stakeholders such as Health Technology Assessment (HTA) bodies, patients, and the U.S. Food and Drug Administration (FDA).
As of December 17, 2025, data published on the EMA website indicate that the EMA received and evaluated over 500 PRIME applications, with a final inclusion rate of 26% into the PRIME scheme. Among these, 26 applications were received in the ophthalmology field, of which 8 were granted inclusion in the program.
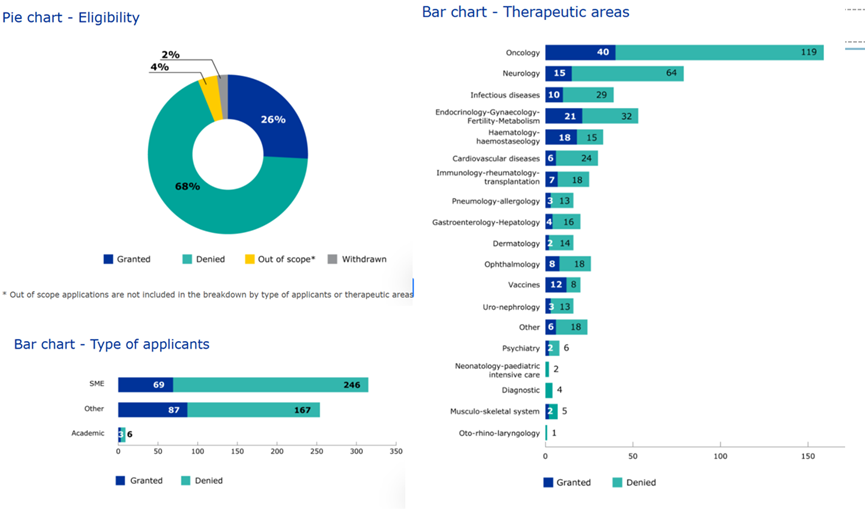
Source: EMA Website
About VGR-R01
VGR-R01 is a gene therapy product independently developed by Vitalgen based on recombinant adeno-associated virus. In November 2022, it was approved by the National Medical Products Administration (NMPA) for use in clinical trials for the treatment of BCD, making it the first globally to enter the registration clinical stage for this indication. Previously, VGR-R01 was included in Phase C of the "Care Plan" by the Center for Drug Evaluation (CDE) of the NMPA in December 2024; it was granted Orphan Drug Designation (ODD) by the Food and Drug Administration in October 2024; and it was approved by the CDE to be included in the Breakthrough Therapy Drug Program in August 2024.